Body Centred Cubic Cell
Spacefill Ball Stick Sticks. 4 Atomic packing factor in body centred cubic unit cell is 68 percent.

Unit Cell Chemistry Atomic Radius Density Edge Length Calculations Unit Cell Nomenclature Chemistry Chemistry
In the body-centered cubic lattice a total 4 radius one complete atom and two half atoms participate at the body.
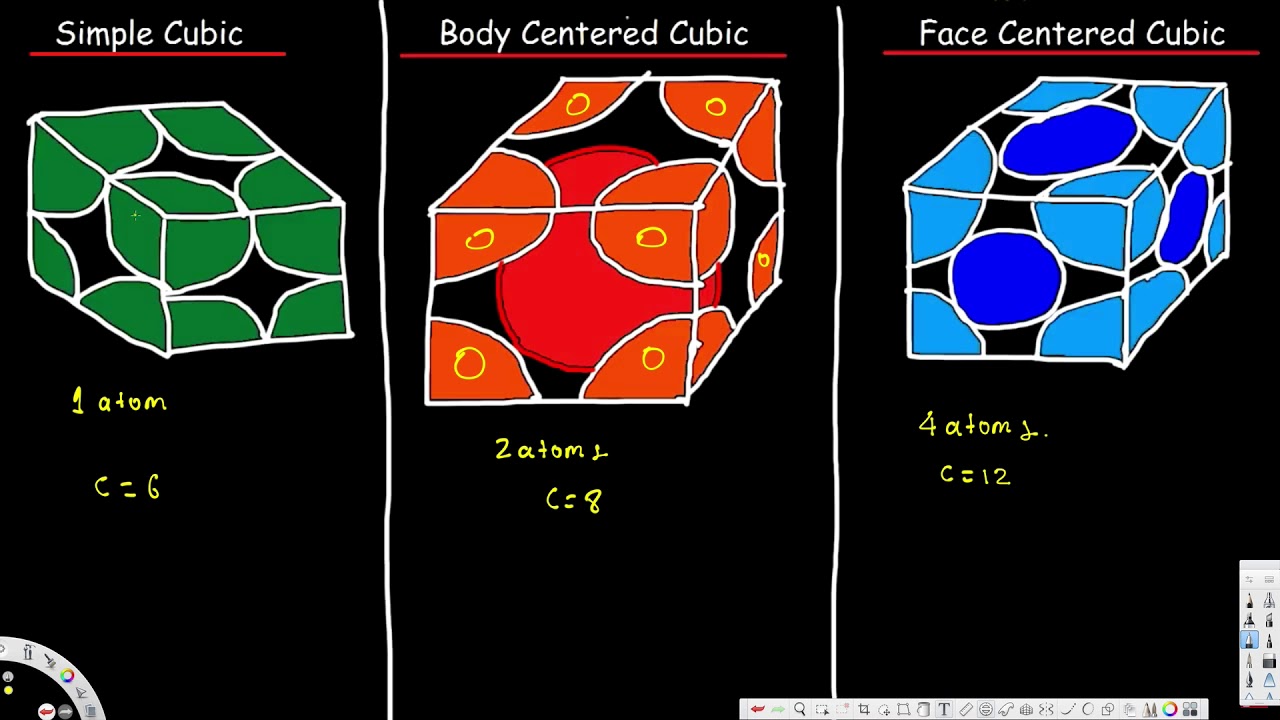
. A body-centered cubic unit cell structure consists of atoms. Body-centred Cubic Unit Cell BCC source. In each cubic unit cell there are 8 atoms at the corners.
A body-centered cubic unit cell structure is composed of atoms organized in a cube with one atom in each corner and one atom in the center. In body centred cubic cell an atom at the body-centre is shared by 1 unit cell. Au 197 gmol A.
How do you measure the edge of a body centered cubic. Naturally occurring gold crystallises in face centred cubic structure and has a density of 193gcm. Eight other unit cells share the atom at the.
Hence the correct option is A. A 533 gcm3 b 429 gcm3 c 571 gcm3 d 052. How do you measure the edge of a body centered cubic.
The body-centered cubic bcc lattice Figure 14b can be obtained by adding a second lattice point at the center of each cubic cell of a simple cubic lattice. Vanadium has a body-centered cubic structure. Thus the unit cell of each bcc.
435 34 votes. 5 Relationship between cube edge length. The body-centered tetragonal unit cell can be imagined as a cube that is slightly taller or shorter in one direction with an atom on each corner and in the very center.
In the body-centered cubic lattice a total 4 radius one complete atom and two half atoms participate at the body diagonal. Atomic packing factor in face-centred cubic unit cell has 12. It crystallizes in a body-centered cubic BCC unit cell.
If the atomic radius of vanadium is 134 pm calculate the density of solid vanadium. This is called a. Europium is the least abundant lanthanide element with symbol Eu atomic number Z 63 and atomic molar mass 16726 g mol 1.
Some metals crystallize in an arrangement that has a cubic unit cell with atoms at all of the corners and an atom in the center as shown in Figure 2. Find the atomic radius of gold. Home Inorganic Chemistry Solid state.
Thus this atom contributes one atom to the unit cell. Frame 1 Next Prev. Therefore the total number of atoms in one unit cell is 8 18 1 atom.
Body-centered cubic BCC is the name given to a type of atom arrangement found in nature.

Unit Cell Simple Cubic Body Centered Cubic Face Centered Cubic Cryst Unit Cell Crystal Lattice Structure Nomenclature Chemistry

Unit Cell Chemistry Atomic Radius Density Edge Length Calculations Unit Cell The Unit Atom

Unit Cell Body Centered Cubic Crystal Lattice Structures Physical E Crystal Lattice Structure Unit Cell Nomenclature Chemistry
11 7 Structure Of Solids Chemistry Libretexts Unit Cell Material Science Physical Chemistry

Cubic Lattice From Wolfram Mathworld Cell Forms Unit Cell Lattice

Planar Packing Fraction Factor For The Body Centred Cubic 111 Plane Fractions Body Packing
0 Response to "Body Centred Cubic Cell"
Post a Comment